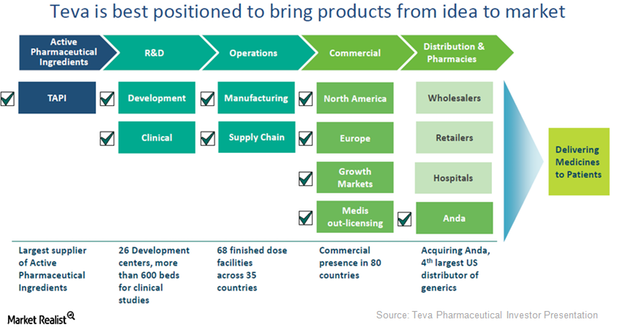
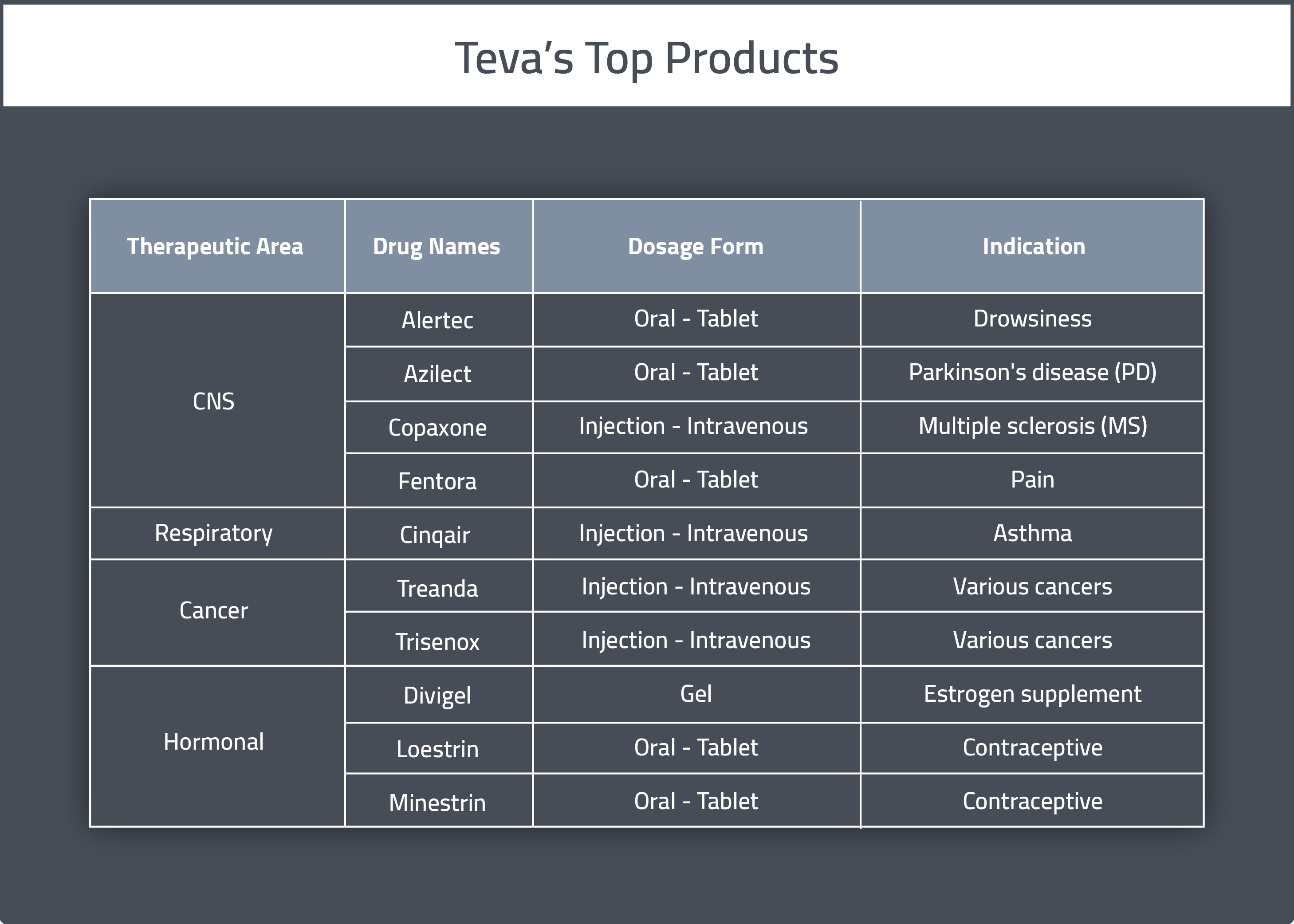
Teva Pharmaceuticals Announces Launch of Daily Maintenance Asthma Medication | Asthma and Allergy Foundation of America

Huntington's disease drug: Teva Pharma settles patent dispute with Lupin, continues to litigate against Aurobindo

PDR0000401 An electronic inhaler add-on module which store and transmit information related to inhaler use (eModule) Cover Letter Letter of Short Term Confidentiality Teva Branded Pharmaceutical Products R&D;, .

Teva Branded Pharmaceutical Products R&D;, . An electronic inhaler add-on module which store and transmit information related to inhaler use (eModule) PDR0000401 FCC ID 2AJVSPDR0000401

Teva Announces New Org Structure, Leadership Changes | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology